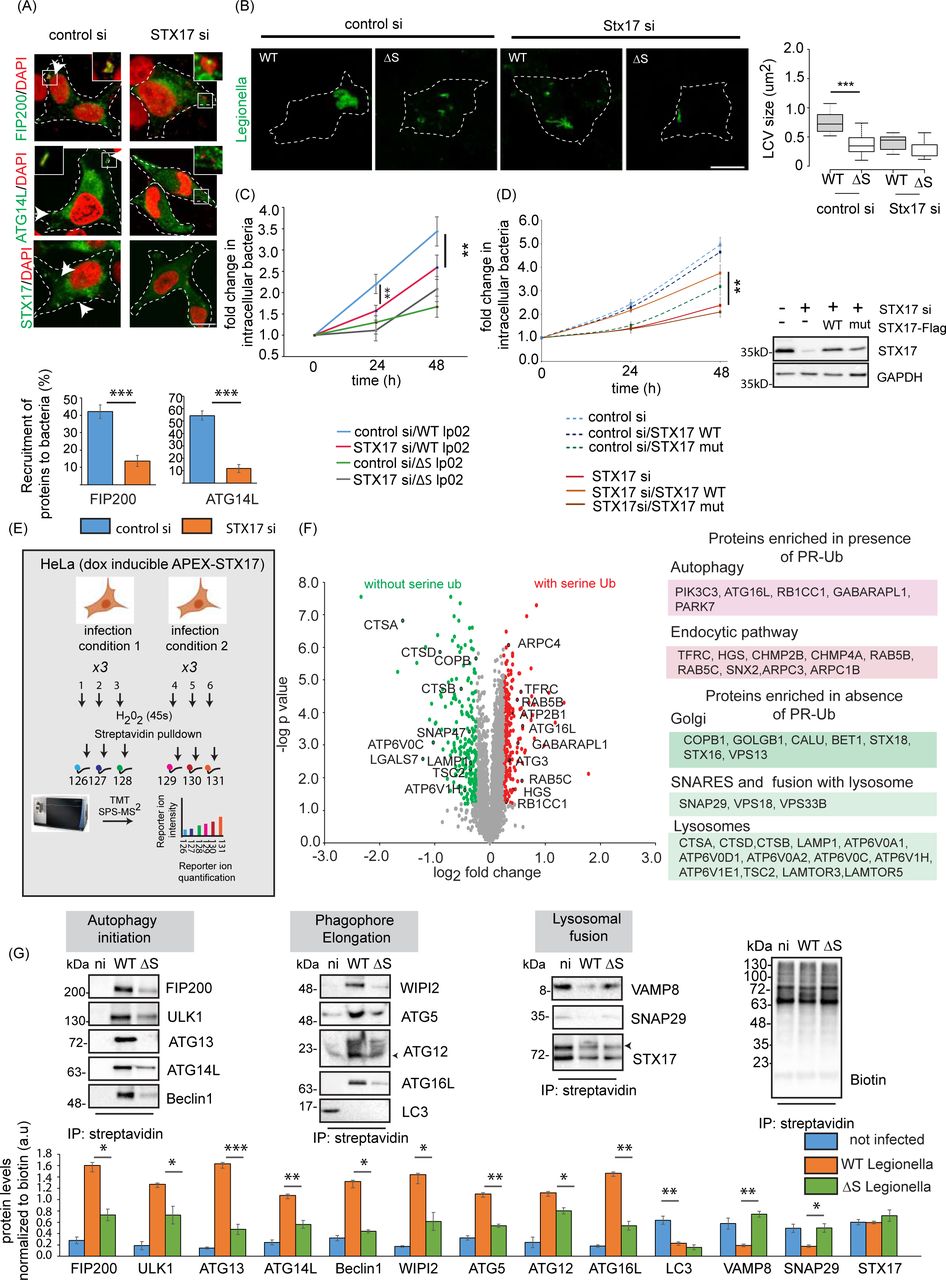
lrp6 (Addgene inc)
Structured Review

Lrp6, supplied by Addgene inc, used in various techniques. Bioz Stars score: 93/100, based on 11 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more
https://www.bioz.com/result/lrp6/product/Addgene inc
Average 93 stars, based on 11 article reviews
Images
1) Product Images from "WNT-induced association of Frizzled and LRP6 is not sufficient for the initiation of WNT/β-catenin signaling"
Article Title: WNT-induced association of Frizzled and LRP6 is not sufficient for the initiation of WNT/β-catenin signaling
Journal: Nature Communications
doi: 10.1038/s41467-025-60096-7

Figure Legend Snippet: A Architecture of LRP-Venus and FZD-Nluc constructs. SP, signal peptide; FLAG, FLAG-tag; E1-E4, extracellular YWTD β-propeller/EGF domain repeats 1-4; LDL-A, low-density lipoprotein receptor type A repeats; TM, transmembrane domain; ICD, intracellular domain; CRD, cysteine-rich domain; 7TM, seven-transmembrane domain; C-term., receptor C-terminal domain. B Schematic depiction of the assay principle. Parts of this figure were created with BioRender.com released under a Creative Commons Attribution-NonCommercial-NoDrivs 4.0 International license. Created in BioRender. Voss, J. https://BioRender.com/lfyvjb8 . C , D . WNT-3A stimulation (1000 ng/ml) kinetics of HEK293 ( C ) or HEK293T ΔLRP5/6 ( D ) cells transfected with FZD 5 -Nluc and LRP5/6-Venus (yellow and blue circles, respectively; note: y-axes are not scaling equal in all figure panels). E Stimulation of HEK293 cells transfected with FZD 5 -Nluc and LRP5- or LRP6-Venus with 1 nM of WNT surrogate. F WNT-3A stimulation (1000 ng/ml) of HEK293 cells transfected with FZD 5 -Nluc and chimeric LRP5/6-Venus variants. G Architecture of LRP6 and LRP6-5A-Venus constructs. PPP(S/T)P/PPPAP, phosphorylation motifs. H. WNT-3A stimulation kinetics of HEK293A cells transfected with FZD 5 -Nluc and LRP6-/LRP6-5A-Venus (blue and light blue circles, respectively). Data are presented as mea n ± standard error of the mean (SEM) of five ( C ), four ( D ), or three ( E , F , H ) individual experiments, each performed in triplicate.
Techniques Used: Construct, FLAG-tag, Transfection, Phospho-proteomics

Figure Legend Snippet: A Schematic depiction of mutational paradigms. The FZD 5 R 6.32 A mutants preferentially signals in DVL-independent fashion. The DVL2 M2/M4 mutant cannot polymerize by its DIX domains. The DVL2 L445E mutant cannot engage FZD via its DEP domain. Created in BioRender. Voss, J. (2025) https://BioRender.com/zfu7r84 . B ΔBRET traces of HEK293 cells transfected either with WT FZD 5 -Nluc or FZD 5 R 6.32 A-Nluc (dark and light blue circles, respectively) and LRP6-Venus, stimulated with 1000 ng/ml WNT-3A. C ΔBRET traces of HEK293 and HEK293T ΔDVL1-3 cells, transfected with FZD 5 -Nluc, LRP6-Venus and either pcDNA (green and black circles), DVL2 (cyan and magenta), or DVL2-M2/M4 (dark cyan and purple) as indicated, stimulated with 1000 ng/ml WNT-3A. D ΔBRET traces of ΔDVL1-3 cells, transfected with FZD 5 -Nluc, LRP6-Venus and either pcDNA (black), DVL1 (green), DVL2 (magenta), or DVL3 (purple) as indicated, stimulated with 1000 ng/ml WNT-3A. E ΔBRET traces of ΔDVL1-3 cells, transfected with FZD 5 -Nluc, LRP6-Venus and either pcDNA (black), DVL2 (magenta), DVL2-M2/M4 (purple), DVL2-L445E (orange), or DVL2-M2/M4-L445E (red) as indicated, stimulated with 1000 ng/ml WNT-3A. F Scheme illustrating a proposed mechanism of DVL modulation of WNT-induced FZD 5 -LRP6 interaction. Created in BioRender. Voss, J. (2025) https://BioRender.com/3gct1ez . Data are shown as mea n ± SEM of three (B – FZD 5 R 6.32 A, C – data with DVL2-M2/M4 co-expression D, E) or four (B – wt FZD 5 , C – all other data) individual experiments performed in triplicate. Parts of this figure were created with BioRender.com released under a Creative Commons Attribution-NonCommercial-NoDrivs 4.0 International license.
Techniques Used: Mutagenesis, Transfection, Expressing

Figure Legend Snippet: A–D Kinetic ΔBRET traces of 1000 ng/ml WNT-3A ( A ), -5A ( B ), -10B ( C ), and -16B ( D ) at FZD 4,5,7 -Nluc (purple, red, and green, respectively) and LRP6-Venus. E Immunoblotting of phospho-LRP6, β-catenin, DVL2 and GAPDH (loading control) from whole cell lysates of HEK cells, stimulated for 2 h with 300 ng/ml of WNT-3A, -5A, -10B, and -16B or 1 nM surrogate WNT. The vehicle control (VC) was treated with HBSS and a CHAPS/EDTA mixture corresponding to that present in WNT preparations. F , G Densitometric analysis ( F , ratio of phospho-LRP6 (P-LRP6)/GAPDH; G , ratio of phosphorylated and shifted (PS-DVL2/DVL2) of blots shown in E. VC – black circle outlines, WNT-3A – blue, WNT-5A – red, WNT-10B – dark teal, WNT-16B –purple, surrogate WNT – gray. H . TOPFlash reporter gene assays in HEK293 and HEK293T ΔFZD 1-10 cells overexpressing FZD 4/5/7 stimulated with diverse WNTs (300 ng/ml for 24 h) or WNT surrogate (1 nM for 24 h). The TOPFlash ratio is given as a fold-increase over vehicle control; statistical analysis was performed with one-way-ANOVA versus vehicle control for each transfection condition. WNT-3A – blue, WNT-5A – red, WNT-10B – dark teal, WNT-16B –purple, surrogate WNT – gray. I . TOPFlash reporter gene assays in HEK293 cells stimulated with R-spondin 1 (RSPO1; 100 ng/ml), WNT-3A (300 ng/ml), WNT-16B (300 ng/ml), and WNT-R-spondin 1 combinations for 24 h. Vehicle – black circle outlines, R-spondin 1 – blue-gray, WNT-3A – blue, WNT-16B – purple, WNT-3A + R-spondin 1 – light blue, WNT-16B + R-spondin 1 – pink. Statistical significance was assessed by a one-way ANOVA using Dunnett’s post hoc test for multiple comparisons against the vehicle control (reporter gene assay data was log 10 -transformed prior to statistical analysis). Data points are shown as mea n ± SEM of three individual experiments ( n = 4 in Fid. 2 C, D for FZD 5 data and in Fig. for FZD 7 TOPFlash data), performed in triplicate in case of BRET assays and reporter gene assays. Exact p-values are detailed in the source data file.
Techniques Used: Western Blot, Control, Transfection, Reporter Gene Assay, Transformation Assay

Figure Legend Snippet: A . Representative single-molecule images showing SNAP-FZD 5 molecules, labeled with SNAP SiR-647 (left) and Halo-LRP6 molecules, labeled with Halo R110 (right). B . Single-molecule trajectory traces of SNAP-FZD 5 (magenta) and Halo-LRP6 (green). C . Proportion of molecular confinement at basal and following 100 nM WNT-3A and 100 nM WNT-16B stimulations. Basal – black, WNT-3A early – light blue, WNT-3A late – blue, WNT-16B early – light purple, WNT-16B late – purple. D . Estimated k on (left) and k off (right) values of FZD 5 -LRP6 interactions at basal and following 100 nM WNT-3A and 100 nM WNT-16B stimulations. Basal – black, WNT-3A early – light blue, WNT-3A late – blue, WNT-16B early – light purple, WNT-16B late – purple. E . Distributions of co-diffusion (blue) and co-confinement (red) events at basal and 100 nM WNT-3A and 100 nM WNT-16B stimulated conditions. F . Representative, dual-color single-molecule images showing SNAP-FZD 5 molecules, labeled with SNAP SiR-647 (magenta) and Halo-LRP6 molecules, labeled with Halo R110 (green) following 100 nM WNT-3A and 100 nM WNT-16B stimulations at late time-point. G . Cluster analysis showing the distributions of photobleaching steps following 100 nM WNT-3A and 100 nM WNT-16B stimulations at late time-point. (C-E) Data points are shown as median ± 95% confidence interval. Early stimulation: 2-10 min, late stimulation: 11-25 min. Statistical comparisons were made by Kruskal-Wallis followed by Dunn’s multiple comparison test. n = 18, 25, 22, 17, 27, 25 cells for FZD 5 -LRP6 basal, WNT-3A early, WNT-3A late, WNT-16B early, WNT-16B late, and β 2 AR-LRP6, respectively, from six independent experiments. See also Supp. Movies - . Exact p-values are detailed in the source data file.
Techniques Used: Labeling, Diffusion-based Assay, Comparison

Figure Legend Snippet: A Proportion of SNAP-FZD 5 and Halo-LRP6-5A molecular confinement at basal (black) and following 100 nM WNT-3A (early – light blue, late – dark blue) stimulation. B Representative, dual-color single-molecule image showing SNAP-FZD 5 molecules, labeled with SNAP SiR-647 (magenta) and Halo-LRP6-5A molecules, labeled with Halo R110 (green) following 100 nM WNT-3A stimulation at late time-point. C . Estimated kon (left) and koff (right) values of FZD 5 -LRP6-5A interactions at basal and following 100 nM WNT-3A stimulation (early – light blue, late – dark blue). D Distributions of co-diffusion (blue) and co-confinement (red) events at basal and 100 nM WNT-3A stimulated conditions. E Cluster analysis showing the distributions of photobleaching steps following 100 nM WNT-3A stimulation at late time-point. A , C , D Data points are shown as median ± 95% confidence interval. Early stimulation: 2-10 min, late stimulation: 11-25 min. Statistical comparisons were made by Kruskal-Wallis followed by Dunn’s multiple comparison test ( A , D ) and Mann-Whitney test C . n = 21, 26, 32 cells for FZD 5 -LRP6-5A basal, WNT-3A early, WNT-3A late, respectively, from three independent experiments. See also Supp. Movies – . Exact p-values are detailed in the source data file.
Techniques Used: Labeling, Diffusion-based Assay, Comparison, MANN-WHITNEY

Figure Legend Snippet: A Direct comparison of the effects of WNT-3A and WNT-16B as probed in this study. B Two-step model of signal initiation and specification in WNT/β-catenin signaling. In a first step, WNT binding leads to ligand-induced association of FZD 5 and LRP6 (signal initiation) for both WNT-3A (top, red) and WNT−16B (bottom, blue). Upon higher-order receptor clustering and LRP6 phosphorylation, the signal is specified into WNT/β-catenin signaling (top, red). If these hallmarks are not met, the FZD-WNT complex can signal via other signaling pathways (bottom, blue). It is unclear whether LRP6 is involved in signal specification in that case. Created in BioRender. Voss, J. (2025) https://BioRender.com/e80oa99 . Parts of this figure were created with BioRender.com released under a Creative Commons Attribution-NonCommercial-NoDrivs 4.0 International license.
Techniques Used: Comparison, Binding Assay, Phospho-proteomics, Protein-Protein interactions